Heat Transfer Lab
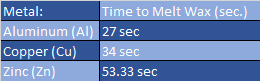
Conduction: At this station you will be observing the difference between how well metal and plastic conduct heat by placing your wrist against either material. Because metals are generally considered to be a better conductor of heat, you compare three different types of metal. Using heat resistant gloves you take the tops of three different metal strips together and place them into a hot bath of water and watch the beads of wax as they melt and slide down the metal strip, record which one passes the 9cm mark first.
A real life example of conduction is if you're cooking at home and the stove. If you accidentally touch the hot stove, then you could burn yourself because you had direct contact with something as hot as that is.
A real life example of conduction is if you're cooking at home and the stove. If you accidentally touch the hot stove, then you could burn yourself because you had direct contact with something as hot as that is.
Convection: For this station you will be observing how convection works. You first need a beaker (specifically 125 mL Erlenmeyer flask) filled with red (you can use food coloring) hot water and put a rubber stopper on top of it, then, using heat resistant gloves or tongs to pick up the hot beaker, then place it in to another beaker (specifically a 1 mL beaker) of cold water and watch what happens.
A real life example of convection in the world is when you're cooking and need to boil water for something. The boiling starts happening because the water at the bottom of the pot heats up and that water rises, only to cool off at the top to sink back down to the bottom and heat up again. All of this is what causes the water to boil.
A real life example of convection in the world is when you're cooking and need to boil water for something. The boiling starts happening because the water at the bottom of the pot heats up and that water rises, only to cool off at the top to sink back down to the bottom and heat up again. All of this is what causes the water to boil.
Convection of a Gas: Next you will be looking at convection of a gas. You need a small dish with some water in it, and a piece of clay with a small candle in it. You will also need a cylinder and a T-shaped divider with foil around the bottom of the T. It should fit snugly in the cylinder. Now you need to light the candle and place the cylinder over the candle. You'll notice that the candle quickly goes out. Take the cylinder off the candle and place the divider in the cylinder and repeat the process of lighting the candle and putting the cylinder over it. Now the candle won't go out because the warm air will rise out of one side of the divider, cool off, and sink back down on the other side of the cylinder, providing the candle with the new oxygen it needs to stay lit.
A real life example of convection of gas in the world would be in the air. There are multiple air currents in the air caused by convection currents with warm air rising and cool air sinking in a circular effect.
A real life example of convection of gas in the world would be in the air. There are multiple air currents in the air caused by convection currents with warm air rising and cool air sinking in a circular effect.
Radiation: Finally you will be observing how radiation works. You need two small beakers, two thermometers, white and black sand, a lamp (specifically with a 100 W bulb), and a timer. Put an equal amount of sand in each beaker and place one thermometer in each. Put both beakers under the lamp and write down what the temperature is for both sand types. Take down the temperature of both types of sand every minute for four minutes. At the end you'll see that the black sand is warmer than the white sand because the black sand absorbed more of the heat that the lamp radiated.
Radiation is practically everywhere. Let's say it was summer and you were at a park and wanted to go down the slide, but it was made out of metal. I wouldn't advise doing so because the sun would have been radiating heat down onto the slide which would have been able to absorb that heat easily.
Radiation is practically everywhere. Let's say it was summer and you were at a park and wanted to go down the slide, but it was made out of metal. I wouldn't advise doing so because the sun would have been radiating heat down onto the slide which would have been able to absorb that heat easily.
Thermal Contact: The state of two or more objects or substances in contact such that it is possible for heat to flow from one object of substance to another.
Specific Heat: The quantity of heat required to raise the temperature of a unit mass of a substance by one degree Celsius.
Insulator: A material that is a poor conductor of heat and that delays the transfer of heat.
Conductor: Material through which heat can be transferred.
Specific Heat: The quantity of heat required to raise the temperature of a unit mass of a substance by one degree Celsius.
Insulator: A material that is a poor conductor of heat and that delays the transfer of heat.
Conductor: Material through which heat can be transferred.